
centrifuge
Centrifugation is the term used to describe a method of separating mixtures using rotation and centrifugal force. Several characteristics of particles can be separated during centrifugation, including size, shape, density, and viscosity.
A centrifuge is a laboratory instrument used for density-based separation of fluids, gases, or liquids.

Laboao centrifuge
The principle of centrifugation is the principle of gravity sedimentation. The rotor of the centrifuge is rotated at high speed to generate a relatively strong centrifugal force, which accelerates the sedimentation of fine particles in the liquid, thereby achieving the separation of the liquid and the fine particles.
Currently, centrifuges are used in chemistry, biology, biochemistry and clinical laboratories, such as testing the sedimentation rate of various blood cells. Centrifugal technology is also used in the production of biological products and APIs, as well as biopharmaceutical analysis of drugs, and is even used in dairy cream (fat) separation, water treatment and other fields.
Centrifuge components
Some common parts of centrifuges are introduced as follows:
1.Motor : The motor is the powerful core component that generates rotation in a centrifuge.
2.Rotor assembly : The drive shaft and rotor make up the rotor assembly. The drive shaft provides support for the rotor components. The rotor head is connected to a motor, which is equipped with a container for holding the test tube containing the sample to be centrifuged. Two rotors of different diameters can have the same rotational speed, and different radii and angular momentum will cause different accelerations of such rotors.
There are three main types of rotors:Fixed-angle rotors, These rotors fix the tube at an angle of 14°~40° to the vertical, allowing the particles to travel short distances while moving radially outward, and are used for differential centrifugation. Since the direction of sedimentation is the same as the direction of centrifugal force, sedimentation occurs at an angle on the pipe wall. The particles (sediment clumps) collide with the wall and then settle at the corners of the base and wall. Swing barrel/horizontal rotor,These rotors swing to a horizontal position together with the centrifuge tube when accelerating, allowing the particles to move a longer distance, thereby facilitating easier separation of the supernatant and pellet for density gradient centrifugation.
Vertical rotor: Holding the tube vertically, that is, parallel to the motor axis, the distance the particles move is shorter and the separation time is shorter. It is used for isopycnic and density gradient separation.
3.container: Various types of containers, such as test tubes, blood bags, cuvettes, centrifuge tubes, etc., are fixed in the rotor so that the sample rotates as the rotor rotates.
4.control Panel : Used to control different parameters, such as temperature, rotation speed (rcf or rpm), etc.
5.latch: The latch keeps the lid closed when a test tube breaks or other problems occur while the centrifuge is running.
6.cover : The centrifuge will only spin when the lid is closed and locked to prevent accidents.
Types of centrifugal technology
There are two types of centrifugation techniques namely preparative centrifugation and analytical centrifugation. Preparative centrifugation involves the separation and purification of components such as tissues, cells, subcellular structures, membrane vesicles, and other biochemically relevant particles. In contrast, analytical centrifugation is performed to characterize purified biomolecules.
Preparative centrifugation
According to the suspension situation, preparative centrifugation is divided into differential centrifugation and density gradient centrifugation.
1. Differential centrifugation
It separates particles based on shape, size and density. Suspensions of particles with different densities or sizes will settle at different rates, with larger and denser particles settling faster. A series of increasing centrifugal force cycles on a cell suspension will produce a series of pellets containing cells with reduced sedimentation rates.

Differential centrifugation
2. Density gradient centrifugation
Separate particles based on their buoyant density or settling rate. The sample mixture is placed on top of a preformed liquid density gradient for DNA banding and separation of plasmids, nucleoproteins and viruses; NaBr and NaI are used for fractionation of lipoproteins; Percoll, Ficoll, Metrizamide, Dextran are used for separation Whole cells, sucrose solution for separation of DNase, RNase and Protease.
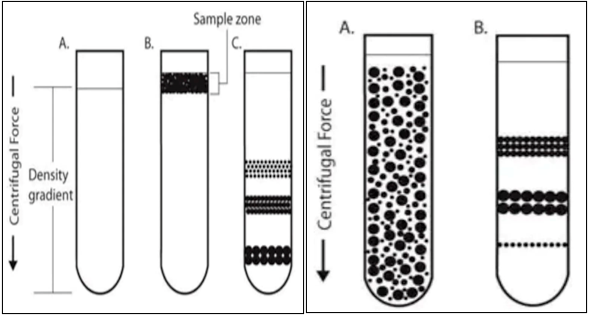
Rate partition centrifugation, isopycnic centrifugation
Density gradient centrifugation is divided into rate zonal centrifugation and isopycnic centrifugation.
Rate zonal centrifugation: The sample is covered as a small area at the top of the density gradient. Depending on their mass, particles travel at different speeds due to centrifugal force. Size and mass are the main determinants of particle settling velocity. As a band of particles descends in a dense medium, regions of comparable sized particles form as faster settling particles pass slower particles.
Isopycnic centrifugation: In isopycnic separation (also called buoyancy separation or equilibrium separation), particles are separated based solely on their density. The gradient medium must have a higher density than the particles that need to be separated. The particles migrate under the influence of centrifugal force and density gradient of the homogeneously mixed sample until their density becomes equal to that of the surrounding medium. After centrifugation, particles of a certain density settle until their density is equal to the density of the gradient medium (i.e., the equilibrium position).
analytical centrifugation
The purpose of analytical centrifugation is to collect information to characterize rotating samples (sedimentation velocity, viscosity, concentration, etc.), determine the relative molecular weight of solutes, the purity of biomolecules, detect conformational changes in protein structure, etc.
Types of centrifuges

1. Desktop or desktop centrifuge
Due to their smaller size, they are very convenient for small laboratories with limited space
These are compact and often used in research and clinical laboratories
Benchtop centrifuge with lid covering the equipment used to run the centrifuge and a rotor with test tube racks
2. Gas centrifuge
They are used to separate molecules based on mass and gases based on isotopes
Specifically, they are used in the extraction and separation of uranium-235 and uranium-238
3. Hematocrit centrifuge
Hematocrit centrifuges operate at speeds from 7000 to 15000 rpm
The main purpose of a hematocrit centrifuge is to calculate the volume-based percentage of red blood cells in the blood. It is used to produce plasma for photometric analysis of bilirubin concentration in neonatal blood.
4. Microcentrifuge
Due to their highly compact form factor, they have a very small footprint and take up minimal space on your workstation
These work well with small tubes (up to 2.0 ml) and are often used in biological applications
They are used to microfiltrate small amounts of water samples and retain precipitated nucleic acids, proteins and other materials in solution.
5. Refrigerated centrifuge
These centrifuges operate at maximum speed while maintaining a constant temperature
It is used to analyze DNA, RNA, PCR and antibodies as its temperature range is between -20 to -40 degrees Celsius
They are often used to quickly collect precipitated materials, including yeast cells, chloroplasts, etc.
6. High speed centrifuge
A high-speed centrifuge is a slightly faster centrifuge, ranging from 15,000 to 30,000 rpm
High-speed centrifuges contain a device that regulates temperature and operating speed for critical analysis of fine biomolecules
These centrifuges use three types of rotors: fixed-angle rotors, bucket rotors, and vertical rotors
7. Low speed centrifuge
These are typically used in laboratories for routine particle sorting operating at a maximum speed of 4000-5000 rpm
Temperature adjustment is rare and usually operates at room temperature
These centrifuges are available in bucket and fixed angle rotor types
8. Continuous flow centrifuge
It can centrifuge large amounts of samples without affecting sedimentation rate
They also have a larger capacity and save time by eliminating the need to repeatedly load and unload samples like standard centrifuges
9. Ultracentrifuge
An ultracentrifuge is a highly developed and sophisticated centrifuge that can separate tiny molecules that cannot be separated quickly by conventional centrifuges
Ultracentrifuge rotor speed range from 60,000 to 150,000 rpm
They run samples as grouped or continuous flow systems and at larger scales
10. Preparative ultracentrifuge
Preparative ultracentrifuges are centrifuges used to separate particles in experiments by centrifugation
When preparing an ultracentrifuge for operation, the contents of the test tube are checked after the centrifugation process, as opposed to a centrifuge used for analysis, where the contents are checked during the centrifugation process.
11. Analytical Ultracentrifuge
Analytical centrifuges are ultracentrifuges used to examine the different particles within a sample
It is used for qualitative examination of macromolecules in solution
They are equipped with sensing devices that track the rotation and movement of ingredients in real time to calculate sedimentation coefficients
Advantages and limitations of centrifuges

Laboao centrifuges
Advantage
Closed operation, neat appearance
Quick startup and shutdown
Can be easily automated and run continuously if necessary
Low capital cost to capacity ratio
Quickly adjust operating parameters
High flexibility and outstanding performance
Simple operation and easy installation
limitations
Separating light particles (with almost negligible mass) is very difficult
Although there are many types of centrifuges, they are very complex electrical equipment that require professional repairs when they malfunction, making equipment extremely difficult and expensive to maintain.
High energy consumption as it has many power-hungry functions such as temperature control and rotor spinning
Due to the high speed per minute, centrifuges can produce noise disturbances. Since it rotates, it inevitably generates vibrations, which can cause noise pollution and hinder its use in some areas.
In most cases, when the primary device fails, a backup machine is required to step in.
Centrifuge operating procedures and precautions

Laboao centrifuges
Operating procedures
1. Check the centrifuge to ensure it is functioning properly, intact and can be moved without restriction.
2. After selecting the appropriate centrifuge tubes or containers, inspect them to ensure there are no flaws or cracks. Any damaged or defective tubes or containers should be discarded.
3. Fill the tube with your preferred liquid, do not overfill or underfill.
4. Check that the centrifuge tubes are balanced; weigh each tube individually on a scale to confirm that they have equal weight. Avoid relying on volume alone for balance! This is especially true for solutions containing various sample types or different sample concentrations.
5. Tighten the cap of the centrifuge tube.
6. Before inserting the centrifuge tube, make sure its outside is dry and clean.
7. Keep the test tubes balanced in the centrifuge.
8. Close the lid and make sure it fits tightly.
9. Configure running time and speed.
10. Do not leave the unit when the centrifuge appears to be running and is running at maximum speed. Check for any unusual vibrations or noises.
11. Once you hear strange sounds or feel strong vibrations, immediately turn off the centrifuge and remove the sample. A typical cause of this is an improperly balanced centrifuge. If the problem does not go away after the centrifuge is properly balanced, do not use the centrifuge until it is repaired.
12. After the centrifuge completes its cycle, wait until it stops spinning before opening the lid. Never touch or open the centrifuge lid until the centrifuge has stopped spinning. Stopping equipment prematurely can result in mechanical failure and injury.
13. In order to allow the aerosols emitted during centrifugation to settle, it is best to wait at least 10 minutes after the rotation stops before opening the lid.
14. Once the centrifuge has stopped spinning, remove the sample from the centrifuge.
Precautions
Before operation, always check that the centrifuge is on a suitable surface
Keep lid closed while rotor is running
When the centrifuge shakes or vibrates, unplug the power plug
Please make sure the tube is suitable for the program and settings before use. Test tubes used in centrifuges should be supplied in complete sets
Pipes should be loaded symmetrically using balanced adjacent and opposing loads. Use mass instead of volume to balance tubes